Industrial hydrogen, grey than green at the moment
This "grey" hydrogen, of fossil origin, is today the cheapest and by far the most widespread manufacturing process: producing one kilogram of hydrogen costs between 1.5 and 2.5 euros, depending on the initial price of natural gas.
How much are the emissions of "grey" hydrogen?
The chemical reaction releases about 10 kilograms of CO2 per 1 kilogram of hydrogen produced, explains Pierre-Etienne Franc, who heads the global hydrogen division of Air Liquide, a French producer of industrial gases, in the book "Hydrogen: the energy transition in motion" (Editions Manifestô).
In France, hydrogen production generates 3% of the country's CO2 emissions and 26% of the country's industrial sector emissions, according to a 2018 government.
What are the "decarbonated" solutions to produce clean hydrogen?
The first path is to capture and store the CO2 emitted during the production of "grey" hydrogen. The injection of CO2 into depleted deposits has an industrial sense: it dissolves the remaining oil and promotes the increase in the volume of this hydrocarbon by decreasing its viscosity.
If you have a side hustle or a course or a group you want to speak about reply to this! Retweet it! Like it! If… https://t.co/yaD2uUtjXn
— mediaboca Wed May 06 18:00:25 +0000 2020
CO2 emissions can also be recycled elsewhere: absorbed by plants in agricultural greenhouses, used as an inert gas for conservation in the agri-food industry, or for the preservation of vaccines at very low temperatures in the pharmaceutical sector. But these solutions remain very marginal.
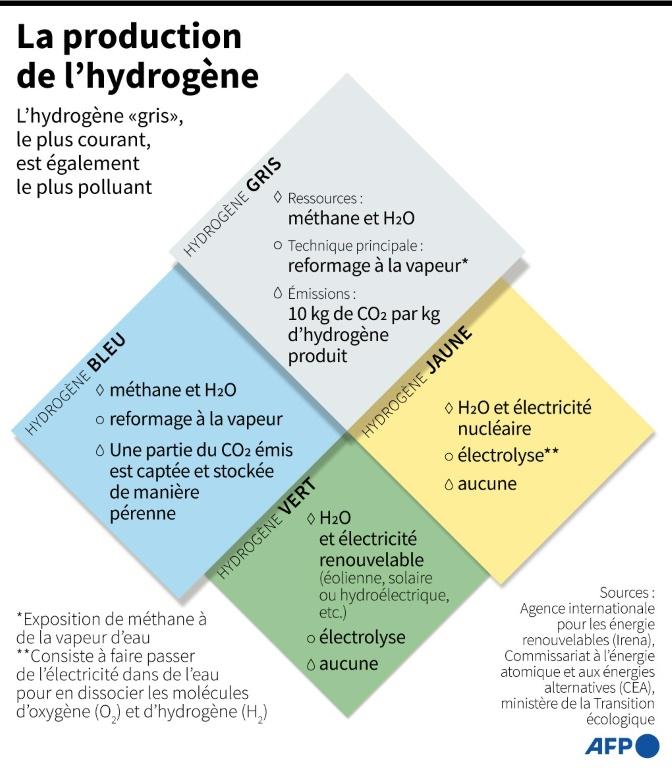
The second approach is to replace fossil natural gas for extraction by biogas resulting from the fermentation of organic waste either from methanisers (France and Europe) or from landfills (United States). The second approach is to replace fossil natural gas for extraction by biogas resulting from the fermentation of organic waste from methanisers (in France and Europe) or from landfills (United States).
There is also a third path, known as "yellow" or "green", through the electrolysis of water, which is also the most expensive. Electricity is passed through water to "break" or dissociate the oxygen (O2) and hydrogen (H2) molecules contained in H2O. This is exactly the opposite of the fuel cell method of assembling hydrogen and oxygen to generate electricity and water.
The advantage of this technique is that there is no release of carbon dioxide. If the electricity used is renewable, wind, solar or hydro-electric, or even biogas, the hydrogen obtained is "green". In the case of nuclear electricity, it is referred to as "yellow".
Currently, hydrogen produced by electrolysis, with yields lower than gas reformers and costs two to four times higher, does not exceed 5% of the world's hydrogen production.
However, it is towards this green hydrogen that investment and research in Germany, France and the United States are being massively directed, in order to create real production channels via electrolysis, in the hope of decarbonizing industry, transport and the economy.
Are there other techniques under consideration?
Pyrolysis could be used to decompose hydrocarbons into carbon and hydrogen. Photosynthesis is also being studied, with some algae naturally producing hydrogen through photosynthesis.